Introduction
Angioimmunoblastic T-cell lymphoma (AITL) is a rare subtype of peripheral T-cell lymphoma derived from T-follicular helper (TFH) cells, as recognized in the 2022 World Health Organization (WHO) Classification of Hematopoietic and Lymphoid Neoplasms. Clinically, AITL is characterized by systemic manifestations, including fever, lymphadenopathy, rash, and immune dysregulation. Up to 30% of patients develop hypergammaglobulinemia, attributed to excessive cytokine secretion by neoplastic TFH cells, which promotes polyclonal B-cell activation and plasma cell differentiation.1
Notably, approximately 20% of AITL cases are associated with a concurrent Epstein–Barr virus (EBV)-driven diffuse large B-cell lymphoma (DLBCL), reflecting the profound immune dysregulation characteristic of this entity.2 In contrast, the development of overt plasma cell neoplasms, such as multiple myeloma (MM), is exceedingly rare and has been reported only in isolated cases (Table 1).
Here, we describe two new patients with AITL complicated by treatment-refractory plasma cell neoplasia, including the first documented instance of primary cutaneous myeloma. We also provide the first comprehensive review of 16 previously published cases with concurrent systemic T-cell lymphoma (TCL) and plasma cell neoplasia (Table 1).
Case Reports
Case 1
A 73-year-old man was admitted in March 2019 with fever, rash, generalized lymphadenopathy, syndrome of inappropriate antidiuretic hormone secretion (SIADH), dry cough, and profound fatigue. Laboratory investigations revealed moderate pancytopenia (neutrophils 0.86×10⁹/L, lymphocytes 0.56×10⁹/L, hemoglobin 9.3 g/dL, platelets 35×10⁹/L) and markedly elevated inflammatory markers (CRP 80 mg/L, ESR 100 mm/h, ferritin 2070 µg/L, LDH 520 U/L, and fibrinogen 580 mg/dL). Serum protein electrophoresis identified a small IgA-kappa paraprotein in the context of marked polyclonal hypergammaglobulinemia (γ-globulin ~40 g/L) and hypoalbuminemia (23 g/L).
Blood and urine cultures, serologies for HBV, HCV, and HIV were negative, but plasma EBV-DNA was elevated at 74,150 copies/mL. Empiric antibiotic therapy was ineffective. Bone marrow aspiration demonstrated trilineage cytopenia with mild dysplastic changes and 10–15% reactive, polytypic plasma cells. PET/CT revealed FDG-avid supradiaphragmatic lymph nodes (up to 2.5 cm) and small bilateral pulmonary nodules. Excisional lymph node biopsy confirmed AITL. Neoplastic T cells expressed CD2, CD3, CD4, CD5, CD10, PD-1, BCL6, CXCL13, and CD30 (10%), were negative for EBER, and had a Ki-67 proliferation index of approximately 40%.
The patient began Cyclophosphamide-Prednisone-Doxorubicin-Vincristine (CHOP) chemotherapy in April 2019, achieving complete remission of systemic symptoms, regression of lymphadenopathy, normalization of blood counts, and decreased gammaglobulin levels (25 g/L). After four cycles, he was re-admitted with fever, severe thrombocytopenia, and epileptic seizures. PET/CT showed only two small residual FDG-avid lung nodules. Bone marrow, brain MRI, and cerebrospinal fluid were unremarkable. High-dose dexamethasone (16 mg/day for several days) led to full recovery and correction of cytopenias.
Brentuximab vedotin was initiated in August 2019, but after three cycles, anemia and thrombocytopenia recurred. In October 2019, treatment was switched to lenalidomide with weekly dexamethasone. By January 2020, rash, fatigue, and pancytopenia reappeared, although PET/CT showed only a single small FDG-avid pulmonary lesion.
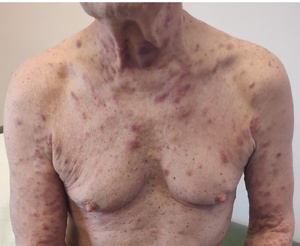
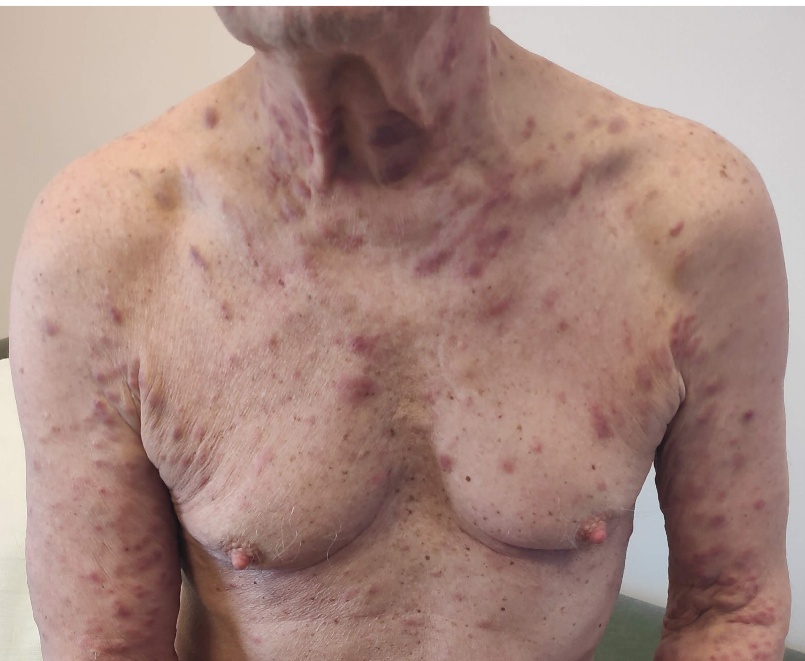
In April 2020, new erythematous plaques developed on the trunk, neck, and upper limbs (Figure 1), coinciding with a sharp increase in the IgA-kappa paraprotein level (35 g/L) and a free light chain ratio >100. EBV-DNA was 68,280 copies/mL. Skin biopsy revealed a dermal infiltrate of clonal plasma cells (CD38+, CD138+, MUM1+, CD56±, IgA-kappa+) consistent with cutaneous myeloma. FISH demonstrated TP53 deletion; Epstein-Barr virus-encoded small RNA (EBER was negative in plasma-cells. Repeat bone marrow biopsy showed no evidence of AITL or myeloma. A new PET/CT scan showed persistence of FDG uptake in the small lung nodule, while no FDG lesions were detected in the bones and other organs. The patient received pomalidomide, bortezomib, and dexamethasone, but the disease rapidly progressed. He developed acute renal failure and died of sepsis in June 2020.
Case 2
In May 2021, a 71-year-old woman presented with night sweats, low-grade fever, and generalized lymphadenopathy. CT imaging revealed multiple enlarged lymph nodes and hepatosplenomegaly. An initial lymph node biopsy showed only reactive changes.
She was referred to our center in June 2021 for further evaluation. Her history included nodular goiter (euthyroid), celiac disease, and resected dermatofibrosarcoma (2016). Laboratory studies showed anemia (hemoglobin 94 g/L), thrombocytopenia (83×10⁹/L), normal leukocytes (5.45×10⁹/L; neutrophils 62%, lymphocytes 30%, monocytes 8%), elevated LDH (350 U/L), and β₂-microglobulin (11.4 mg/L). Serum immunoglobulins (Ig) revealed marked polyclonal hypergammaglobulinemia with elevated IgA and IgG levels. Serology showed isolated anti-HBc positivity, consistent with past HBV infection.
Bone marrow examination demonstrated a 10% infiltrate of CD3+ T cells and 20% polytypic CD138+ plasma cells, with scattered EBV-positive cells by EBER in situ hybridization. Peripheral blood flow cytometry was non-diagnostic. Plasma EBV PCR was 247,000 copies/mL. PET/CT (August 2021) demonstrated hypermetabolic lymphadenopathy and splenic involvement (SUVmax 7.4). An excisional axillary lymph node biopsy confirmed EBV-positive AITL (CD2+, CD3+, CD5+, CD4+, PD-1+, EBER+) coexisting with EBV-positive DLBCL (CD20+, CD79a+, CD10+, EBER+), with a Ki-67 index of 75%. The composite lymphoma was staged as III-B.
Given her age and dual pathology, R-GEMOX chemotherapy (rituximab, gemcitabine, oxaliplatin) was initiated in September 2021. After one cycle, EBV-DNA became undetectable, but following the second cycle, she developed Staphylococcus aureus sepsis and Escherichia coli urinary infection, with EBV reactivation (72,800 copies/mL). PET/CT showed persistent lymphadenopathy, new bone lesions, ascites, and pleural effusion. Therapy was held until recovery.
In January 2022, lymph node biopsy confirmed persistent AITL. Rituximab-Cyclophosphamide-Liposomal Doxorubicin-Prednisolone-Vincristine therapy was commenced, leading to gradual clinical improvement and a decline in EBV viremia (7860 cp/mL). She completed six cycles by April 2022 without major toxicities. In April, after four cycles of RGEMOX, she achieved complete remission by PET-CT scan, EBV viremia was 3780 cp/mL, hemoglobin and platelets normalized, although a mild lymphocytosis (~5×10⁹/L) was noted.
In May 2022, she experienced an abrupt clinical decline with severe anemia (Hb 8.6 g/dL) and extreme leukocytosis (138.5×10⁹/L). Peripheral blood smear and flow cytometry showed 77% circulating plasma cells (CD38+, CD138+, CD56±, CD20±, CD19−), diagnostic of primary plasma cell leukemia (pPCL). Serum studies demonstrated an IgG-lambda monoclonal protein, FLC λ 15.11 mg/dL, FLC κ 6.19 mg/dL (κ/λ ratio 0.41), and λ Bence Jones proteinuria. β₂-microglobulin was 10.8 mg/L, albumin 3.6 g/dL, renal function and calcemic level were within normal ranges (ISS stage III). LDH was 7500, and EBV peaked at 4.977.500 cp/mL, but plasmablasts were not analyzed for EBV-DNA integration. She was hospitalized and treated with plasmapheresis, high-dose dexamethasone, and bortezomib, but her white blood cell count continued to rise, peaking at 203×10⁹/L. Despite aggressive management, she developed septic shock and died shortly thereafter.
Discussion
We report two new patients with concurrent angioimmunoblastic T-cell lymphoma (AITL) and plasma cell neoplasia, along with the first comprehensive review of similar cases published to date. One of our patients represents the first documented case of primary cutaneous myeloma (Figure 1). In fact, cutaneous involvement by myeloma is exceedingly rare (1–2%), and typically occurs in advanced-stage disease as a secondary localization.3 The second patient developed a primary plasma-cell leukemia. In both individuals, the plasma-cell dyscrasia followed an aggressive, treatment-refractory course, leading to death within two months from diagnosis of both malignancies.
T-cell lymphomas (TCLs) and multiple myeloma (MM) originate from distinct lymphoid lineages, and their coexistence in a single patient has been only anecdotally reported. To date, 18 cases—including the two described herein—have been documented. Their median age (70.5 years) and male-to-female ratio did not differ from those typically reported in TCL. These cases involved several TCL subtypes (Table 1): seven AITL, five peripheral T-cell lymphomas not otherwise specified (PTCL-NOS), four ALK-negative anaplastic large-cell lymphomas (ALCL), and two adult T-cell leukemia/lymphomas (ATL). This finding was rather unexpected, as plasma-cell proliferations are typically associated with lymphomas derived from follicular helper TFH cells. Indeed, most TCL subtypes secrete cytokines that profoundly disrupt immune regulation and may promote the uncontrolled proliferation of other immune cell populations.4–6
Similarly, in T-cell large granular lymphocytic leukemia (T-LGLL)—a chronic T-cell neoplasm—associations with monoclonal gammopathy of undetermined significance (MGUS) is frequently observed, and even MM has been reported. However, in contrast to TCLs, both T-LGLL and plasma-cell dyscrasias usually follow an indolent course.7–9
In most cases of this series, the plasma-cell neoplasia was diagnosed synchronously or a few months after TCL (Table 1), suggesting that their co-occurrence is unlikely to be coincidental. Interestingly, the distribution of monoclonal protein isotypes in these patients differs from that in typical MM: IgA was the most frequent (~50%), followed by IgG (~17%), IgM and light-chain types (~12% each), and IgD (5%). Four out of 18 patients developed primary plasma-cell leukemia, and in five who had FISH analysis of plasma-cells, this showed high-risk cytogenetic aberrations, including TP53 deletion in four of them (Table S1). The overall outcomes were poor, as the median survival after diagnosis of the second malignancy was less than two months (range, 1–13 months). Deaths were mainly attributable to rapid disease progression, refractoriness to therapy, and infectious complications.
Several mechanisms may concur with the development of plasma-cell neoplasm in TCL:
1. Profound immunosuppression in TCLs, particularly AITL, facilitates Epstein–Barr virus (EBV) reactivation in B cells. In fact, most patients exhibit high plasmatic titers of EBV-DNA, which typically decline during remission, as in the two new cases herein reported. EBV-transformed B-cell clones may undergo malignant transformation; indeed, at diagnosis, up to 20% of AITL cases harbor a concurrent EBV-related diffuse large B-cell lymphoma (DLBCL).2 Conversely, in our series, EBV was detected in neoplastic plasma cells in only a minority of cases, suggesting that alternative pathogenic mechanisms may be predominant in the concurrent development of TCL and MM. Indeed, EBV-negative DLBCLs with marked plasmocytic differentiation—ranging from mature plasma cell proliferations to plasmolysis lymphoma—have also been described in AITL.10 However, EBV reactivation induces immune dysregulation, potentially contributing to the development of MM.11
2. Immune stimulation driven by cytokine dysregulation from neoplastic T cells4,6 — particularly those of TFH origin — and sustained antigenic stimulation within the tumor microenvironment may promote aberrant B-cell activation and plasma cell differentiation.5 In fact, anecdotal cases of extreme polyclonal plasmacytosis have also been described in AITL.12,13 Several cytokines secreted by malignant T cells, including IL-5, IL-6, IL-21, and TGF-β1, can promote B-cell survival, differentiation, and class switching—most notably toward IgA production.14,15 This cytokine milieu may account for the high frequency of IgA-type MM observed in this series. (Table 1).
3. Direct cellular interactions between neoplastic T cells and plasma cells, which have been occasionally observed histologically. Magro et al. reported that both AITL and cutaneous T-cell lymphomas may be infiltrated by clonal plasma cells.16,17 Such close cell-to-cell interactions could promote reciprocal growth of both tumor populations. Indeed, plasmablasts produce high amounts of IL-6, which is also pivotal for inducing the TFH differentiation program18
4. A common hematopoietic progenitor that acquires distinct oncogenic mutations may lead to divergent clonal evolution. In fact, AITL tumorigenesis is a multistep process, and premalignant cells may also differentiate into tumor-infiltrating B cells that ultimately mature into Ig-secreting plasma cells. Indeed, Fujisawa and collaborators19 showed that TET2 and DNMT3A mutations were present in both T-lineage tumor cells and infiltrating clonal B cells in the majority of analyzed patients.
5. A lineage switch may also be hypothesized.20 In fact, rearrangements of Ig genes as well as T-cell receptor genes have been reported in up to 40% of AITL samples.21 Unfortunately, in the two cases here described, TCR and Ig gene rearrangements were not assessed in either the MM or the T-lymphoma cells. However, lineage shift or a common progenitor origin was ruled out by molecular analyses in a few cases from this series.17,22
6. Previous therapies—including corticosteroids, immunomodulatory agents, and cytotoxic drugs—may also alter immune surveillance and the bone marrow microenvironment, predisposing to secondary neoplasms.
Further studies are needed to elucidate the biological mechanisms underlying the co-occurrence of TCL and MM, which may be underreported and associated with a dismal prognosis. Indeed, such patients should be considered for alternative or targeted therapies, such as anti-interleukin-6 antibodies, to disrupt potential synergism between neoplastic T cells and neoplastic plasma cells. Finally, we recommend that patients with TCL who present with hypergammaglobulinemia or paraproteinemia be thoroughly assessed and longitudinally monitored to rule out this rare but clinically significant association.
Authors’ Contribution
Conceptualization MCC
Data curation CS, EG, GMP
Formal Analysis MCC, CS
Funding acquisition
Supervision MP, AV
Writing – original draft CS, GMP
Writing – review & editing MCC, CS, EG, GMP, AZ, LP, LF, MP, AV
Competition of Interest – COPE
None of the Authors has competing interests to be disclosed
Informed Consent Statement and Ethics approval
Informed consent was obtained from both patients. All authors and institutions have approved this manuscript for publication. Our institution does not require ethical approval for case series.
Data Availability Statement
All are available upon reasonable request.